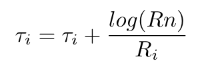
Syntax:
surf_react ID adsorb model infile(s) Nsync type temp n_sites adsp1 adsp2 ...
gs = gas-surface reactions ps = pure-surface reactions gs/ps = both gas-surface and pure-surface reactions
one file for model gs or ps two files for model gs/ps, gs file first, ps file second
face = domain boundary treated as a surface surf = surface elements = triangles in 3d, lines in 2d
Examples:
surf_react adsorb gs gs_react.surf nsync 10 surf 1000 6.022e18 O CO surf_react adsorb gs/ps gs_react.surf ps_react.surf nsync 1 face 300 3e9 O
Description:
Define a model for surface chemistry reactions to perform when particles collide with surface elements or the global boundaries (faces) of the simulation box, which enables the particles to adsorb to the surface. This model also has an option to encode chemical reactions that can occur on the surface itself.
One or more models can be defined and assigned to different surfaces or simulation box boundaries via the surf_modify or bound_modify commands. See Section 6.9 for more details of how SPARTA defines surfaces as collections of geometric elements, triangles in 3d and line segments in 2d. Also see the react command for specification of a gas-phase chemistry reaction model.
The per-surface element species state computed and updated by this command can be output via the dump surf command, using the s_name syntax to output any of the 5 custom surface state variables created by this surface reaction model. They are as follows:
See the examples/surf_react_adsorb dir for scripts that use this surface reaction model.
The ID for a surface reaction model is used to identify it in other commands. Each surface reaction model ID must be unique. The ID can only contain alphanumeric characters and underscores.
In this adsorb style, gas particles can adsorb on the surface. Adsorbed particles can then undergo reactions with other adsorbed particles as well as with new gas-phase particles that strike the surface. Each surface element stores its "state" for the counts of different particle species currently adsorbed on the element, which alters the probablity for future reactions to take place.
When a particle adsorbs on the surface, a gas particle is deleted and the number of adsorbed particles of that particular species is incremented. When a particle desorbs from the surface, a new gas particle is created and the surface count is decremented. The exact location of the adsorbed particles is not stored; they are assumed to uniformly distributed across the surface element.
One of three options is specified by the model keyword: gs or ps or gs/ps. This specifies one of two types of reactions to perform, or both in the gs/ps case.
Gas-surface (GS) reactions involve both gas-phase and adsorbed/bulk species as reactants. Hence GS reactions occur when gas particles collide with the surface. Pure-surface (PS) reactions involve only adsorbed/bulk reactants and do not include any gas-phase atoms or molecules. They are performed once every Nsync timesteps based on the current surface coverage of the various adsorbed species.
For GS reactions, when a particle collides with a surface element or boundary, the list of all reactions possible with that species as the reactant is looped over. A probability for each reaction is calculated, using the formulas discussed below, and a random number is used to decide which reaction (if any) takes place. A check is made that the sum of probabilities for all possible reactions is <= 1.0, which should normally be the case if reasonable reaction coefficients are defined.
For PS reactions, a time counter algorithm is used. Each reaction has a time counter tau that is increased by a value of nsync*timestep if all the reactants are available. The rate for each reaction R is computed based on the specified reaction rate constants as well as the surface coverage of the reactants. The product of the rate R and time counter tau is normalized to obtain the probability for each of reaction; and a random number is used to decide which reaction occurs. The chosen reaction is perforemd and its time counter is modified as follows:
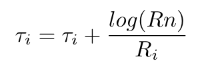
Next the probabilities of each reaction are recomputed and a new reaction is chosen based on these probabilties and performed. This process is repeated till the probability of all the reactions reaches zero. A detailed description can be found in Swaminathan Gopalan et al. (SG18).
The infile argument(s) specify one or two filenames which contain a list of GS or PS reactions and their associated parameters. Two files must be specified for the gs/ps model: a GS file first and a PS file second.
The reactions are read into SPARTA and stored as a list. Each time a simulation is run the list is scanned, and only active reactions are flagged. In order for a reaction to be active, all the reactants and all the products must be currently defined as species-IDs; and also all the surface reactants and products must be present within the list of species that are allowed to adsorb on the surface. The list of surface species is provided at the end of the surf_react adsorb command via the adsp1, adsp2, etc arguments.
Note that this means the reaction file(s) can contain more reactions than are used in a particular simulation.
As explained below each reaction has a specified probability between 0.0 and 1.0. That probability is used to choose which reaction (if any) is performed.
The format of either a GS or PS reaction file is as follows. Comments or blank lines are allowed in the file. Comment lines start with a "#" character. All other entries must come in 2 or more lines with values separated by whitespace in the following format
R1(g) + R2(s) + R3(b) --> P1(g) + P2(s) + P3(b) reaction-type reaction-style C1 C2 ... scattering-model args
The first line is a text-based description of a single reaction. R1, R2, and R3 are the reactants; while P1, P2, and P3 are the products listed as a species IDs. The phase of the reactants and products are species in the brackets immediately following the species ID (no space). It can be one of the following
(g) - gas phase = gas particle striking or scattering from the surface (s) - surface phase = adsorbed particle present on the surface (b) - bulk phase = material which the surface is made of
For a GS reaction, R1 must be a gas-phase reactant. I.e. the particle species that collides with the surface/boundary. For a PS reaction, R1 must be an adsorbed or bulk-phase species.
Individual reactants and products must be separated by whitespace and a "+" sign. The left-hand and right-hand sides of the equation must be separated by whitespace and "-->". The type of each reaction is a string of characters (upper or lower case). The different types of GS and PS reactions are described below.
The allowed types for GS reactions is as follows:
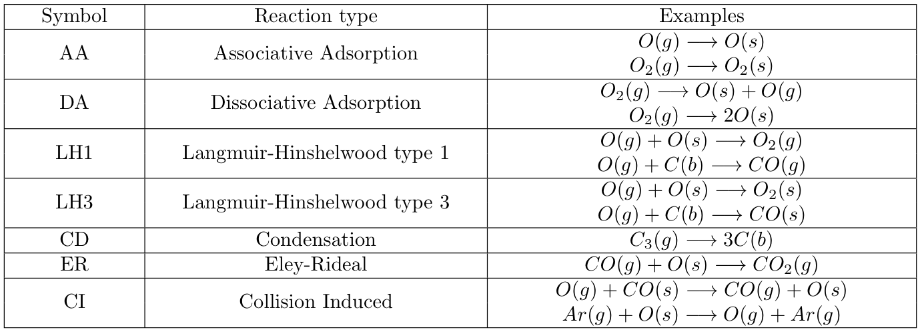
An associative adsorption (AA) reaction means that R1(g) adsorbs on the surface to form P1(s) when it collides with the surface/boundary.
A dissociative adsorption (DA) reaction means that R1(g) dissociates when it collides with the surface/boundary. One or more of the dissociated products can adsorb on the surface and the rest of them scatter as gas-phase products.
A Langmuir-Hinshelwood reaction of type 1 (LH1) means that R1(g) adsorbs on the surface when it collides with the surface/boundary, and reacts quickly with the adsorbed species R2(s) or bulk-phase species R2(b) on the surface to form gas-phase product P1(g).
A Langmuir-Hinshelwood reaction of type 3 (LH3) means that R1(g) adsorbs on the surface when it collides with the surface/boundary, and reacts quickly with the adsorbed species R2(s) or bulk-phase species R2(b) on the surface to form adsorbed product P1(s).
A condensation reaction (CR) means that R1(g) coalesces with the bulk material with which the surface is made of, when it collides with the surface/boundary to form bulk-phase product P1(b).
An Eley-Rideal (ER) reaction means that R1(g) reacts quickly with adsorbed species R2(s) or bulk species R2(b) when it collides with the surface/boundary to form gas-phase product P1(g).
A collision-induced (CI) reaction means that R1(g) causes the desorption of adsorbed species R2(s) when it collides with the surface/boundary to form gas-phase products P1(g) (same species as R2). The incident gas-phase particle can either adsorb P2(s) or scatter from the surface P2(g) post collision.
A detailed description of the various types of reactions can be found in Swaminathan Gopalan et al. (SG18). Here is a table of examples for each type of GS reaction.
The allowed types for PS reactions is as follows:
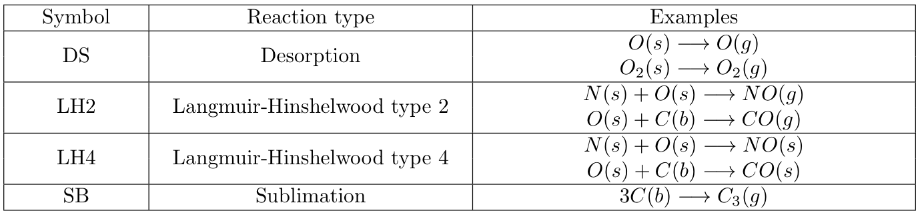
A desorption reaction (DR) means that R1(s) desorbs from the surface to form P1(g) whose final velocities are determined by the provided scattering model.
A Langmuir-Hinshelwood reaction of type 2 (LH2) means that R1(s) adsorbed on the surface reacts with another adsorbed species R2(s) or bulk-phase species R2(b) on the surface to form gas-phase product P1(g).
A Langmuir-Hinshelwood reaction of type 4 (LH4) means that R1(s) adsorbed on the surface reacts with another adsorbed species R2(s) or bulk-phase species R2(b) on the surface to form adsorbed product P1(s).
A sublimation reaction (SR) means that R1(b), the bulk material undergoes a transformation from solid to gas, to form gas-phase product P1(g).
A detailed description of the various types of reactions can be found in Swaminathan Gopalan et al. (SG18). Here is a table of examples for each type of PS reaction.
The style of each reaction is a single character (upper or lower case) with the following meaning:
IMPORTANT: The style of the reaction determines how many reaction coefficients are listed as C1, C2, etc, and how they are interpreted by SPARTA.
For S = Simple style, there is a single coefficient:
C1 = direct value for the reaction rate constant
For A = Arrhenius style, there are three coefficients:
A = pre-expoential factor b = temperature exponent Ea = activation energy for the reaction
The reaction rate constant is calculated in the following manner:
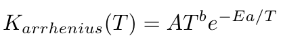
For all the reactions types which includes adsorption - AA, DA, LH1, LH3, and CD; the user must specify the number of species that adsorb on the surface for the reaction as a argument after the reaction rate coefficients
Additional optional keywords for GS reactions can be used to define the reaction rate constant. These are kisliuk: proposed by Kisliuk (Kisliuk57); and energy: proposed by Beckerle et al. (Beckerle89).
kisliuk args = A_k B_k Ea_k (only for reactions which includes adsorption - AA, DA, LH1, LH3, and CD)A_k = pre-expoential factor B_k = temperature exponent Ea_k = activation energy for the adsorption
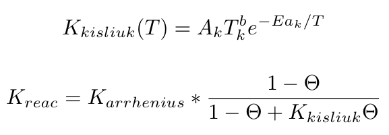
m = energy exponent n = polar angle exponent
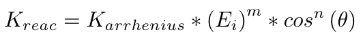
The final rate of the reaction is computed by the product of the reaction rate constant and the surface coverage of all the adsorbed reactants. The reaction probability is obtained by normalized all the reaction rates. A detailed description can be found in Swaminathan Gopalan et al. (SG18).
If there are gas-phase products, there is an option to specify the model used to compute how the particle(s) scatter from the surface for this reaction. This will override the surface collision model assigned to the surface element using the surf_collide command. If no reaction-specific scattering model is desired, specify a NULL value.
Any of the following surface collision models can be used: specular, diffuse, adiabatic, cll, impulsive, td. The scattering model style and its corresponding arguments are specified in the line following the reaction-style. If there are two gas-phase products, two lines (for the first and second particle) can be specified. The arguments for the different surface scattering models are the same as specified in the surf_collide command.
Output info:
All the surface reaction models calculate a global vector of values. The values can be used by the stats_style command and by variables that define formulas. The latter means they can be used by any command that uses a variable as input, e.g. the fix ave/time command. See Section 4.4 for an overview of SPARTA output options.
This adsorb style compute a vector of length 2 + 2*nlist. Nlist is the sum of both the GS and PS reactions listed in the file(s) read as input. Note that this count includes all reactions in the files, not just the ones flagged as active for a particular simulation.
The first element of the vector is the count of particles that performed surface reactions for surface elements assigned to this reaction model during the current timestep. The second element is the cummulative count of particles that have performed reactions since the beginning of the current run. The next nlist elements are the count of each individual reaction that occurred during the current timestep. The final nlist elements are the cummulative count of each individual reaction since the beginning of the current run.
Restrictions:
If the following conditions are met:
then the timesteps on which balancing or grid adaptation are performed must be multiples of Nsync. This is because surfaces are re-assigned to processors due to the change in the grid assignment to processors and the per-surface tau values must be upated appropriately when that occurs.
Related commands:
surf_react, react, surf_modify, bound_modify,
Default: none
(SG18) K. Swaminathan Gopalan, "Development of a detailed surface chemistry framework in DSMC", AIAA Aerospace Sciences Meeting, Jan (2018).
(Kisliuk57) P. Kisliuk, "The sticking probabilities of gases chemisorbed on the surfaces of solids", Journal of Physics and Chemistry of Solids, vol. 3, no. 1-2, pp. 95-101, 1957.
(Beckerle89) J. Beckerle, A. Johnson, and S. Ceyer, "Observation and mechanism of collision-induced desorption: CH4 on Ni (111)", Physical Review Letters, vol. 62, no. 6, p. 685, 1989.